Notice
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Debate Session 4 : Treatment optimization (William T. ABRAHAM)
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
MODIGLIANI Debate Session 4 - Saturday December 1st, 2012:
HEART FAILURE REMOTE MONITORING TRIALS
Chairpersons: Stefan ANKER, Berlin, GER - Ileana PIÑA, New York, USA
Webcast: Daniela DOBRE, Nancy, FRA
Case studies and ultimate methodology for future trials
Remote monitoring remains an appealing option for following patients after a heart-failure hospitalization. Trials employing common strategies (simple phone calls with a nurse and Telemonitoring of vital signs) have in general not demonstrated improvement in survival and or reduction in risk of readmission; meta-analysis of telemonitoring trials using these common strategies, however, have shown improvements in survival and hospitalization rates. Recently, trials of more complex implanted remote monitoring devices that provide physiologic information have reported mixed results.
The 2 most recently published device remote monitoring trials are excellent case-studies that may serve for methodologically refining future trials:
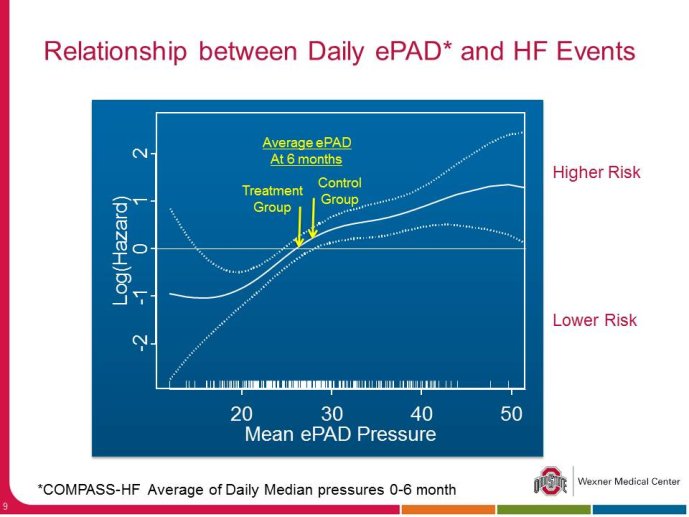
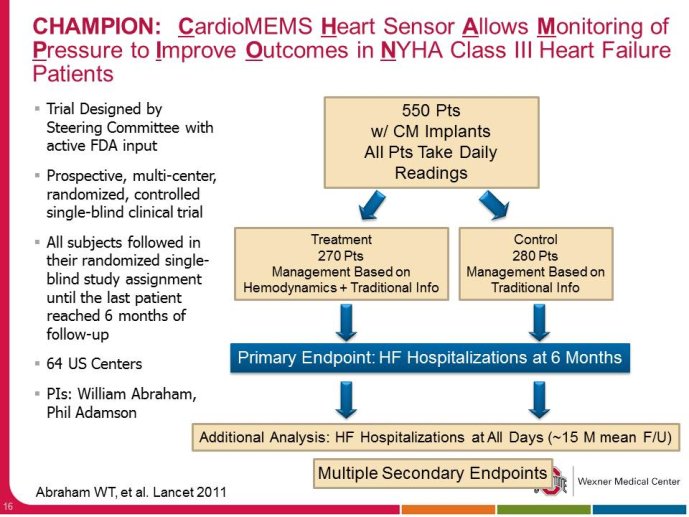
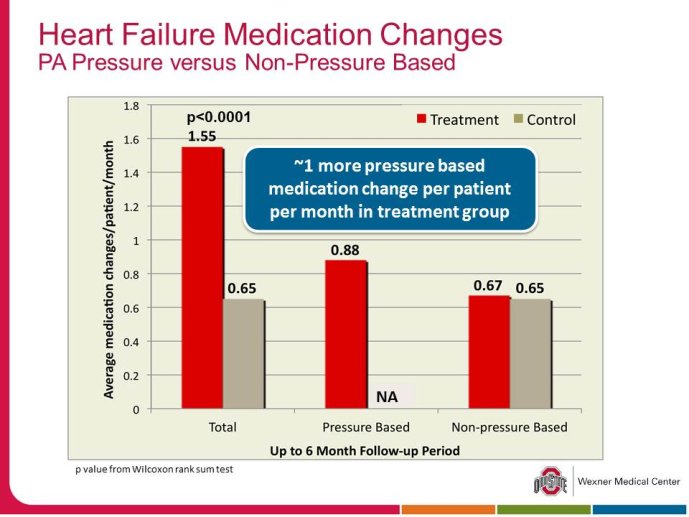
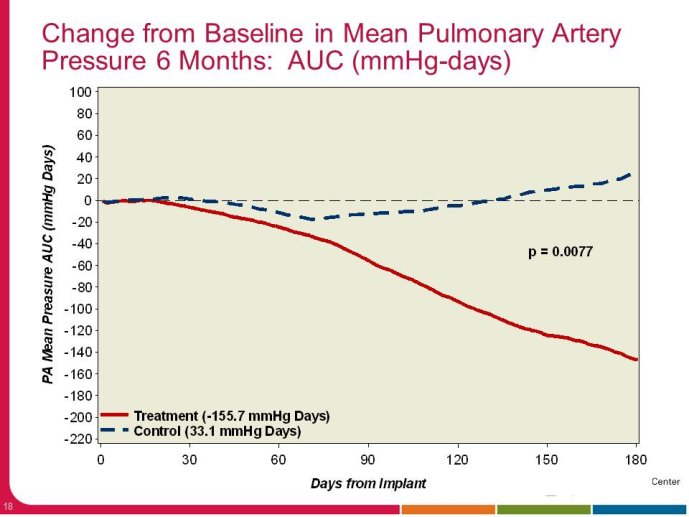
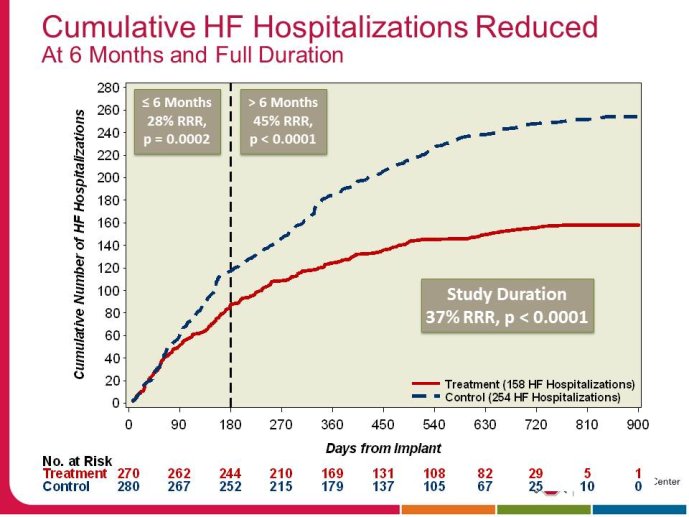
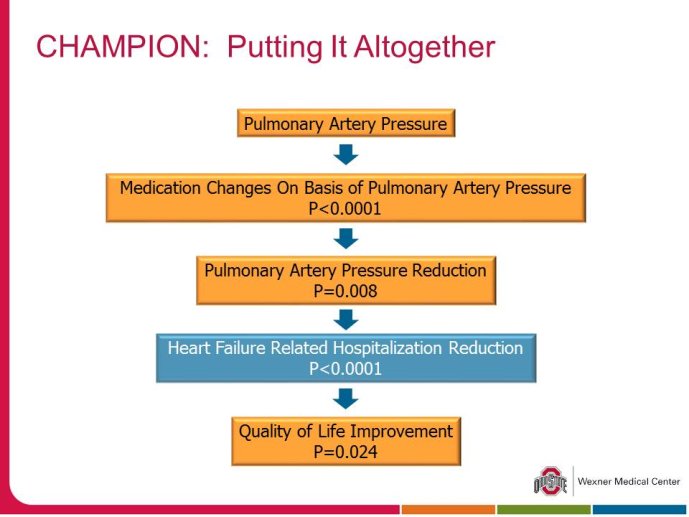
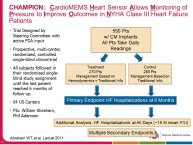
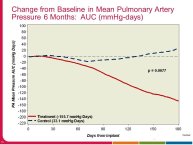
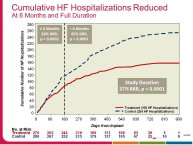
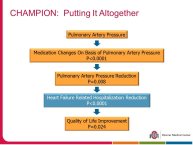
• The CHAMPION trial was the first trial of a technology based (pulmonary artery pressure) telemonitoring (CardioMEMS), published in a major scientific journal (The Lancet) without standing positive results. The FDA advisory panel found the device to be safe (9-1 vote) but did not vote in favor of efficacy (4-6 vote) because of inability to distinguish the effect of the device from the support provided by the Sponsor to the investigators during the trial. The panel and the FDA were also concerned that this level of Sponsor support may not be practical in the commercial setting. The PIs and the Sponsor, however, pointed out that the device provides information only and to adequately test the trial hypothesis (management of pulmonary artery pressure in addition to routine HF care would reduce HF hospitalizations), there had to be a protocol compliance mechanism to ensure that physicians reviewed and responded to pulmonary artery pressures
per the protocol mandated guidelines. This trial poses interesting methodological questions regarding the evaluation of diagnostic devices; how do we design trials with therapeutic endpoints for purely diagnostic devices?
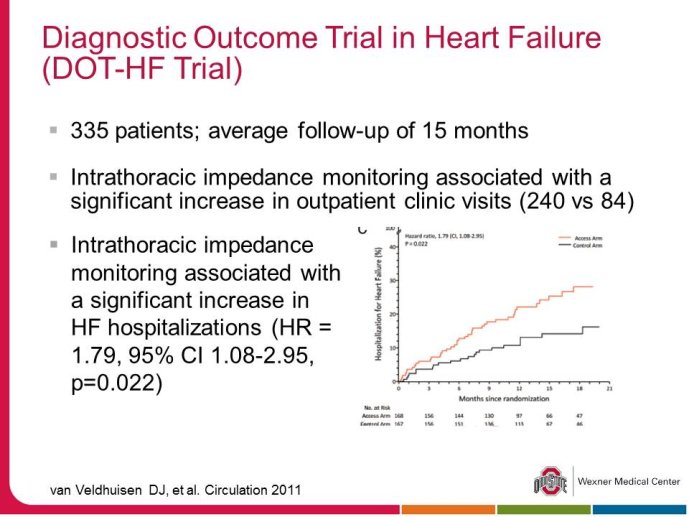
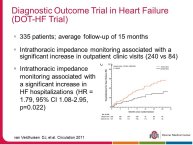
• The DOT-HF trial reported that Telemonitoring based on thoracic impedance was unexpectedly associated with more frequent hospital admissions in the Telemonitoring group. Then the question arises whether this trial failed because the technology - i.e. impedance - failed to deliver the right alerts at the right time or whether the strategy of care using the technology (beep alerts to the patient, no decisio support to the health care providers who managed patients according to a standardized intervention algorithm, on the basis of the available data and the clinical evaluation) was inadequate/ insufficient.
The aim of this workshop is by analyzing lessons from these 2 case-studies, to hopefully reach agreement on the ultimate methodology of future trials of remote monitoring.
Session program:
How to minimize bias? Trial design, control group, blinding and other methodological issues
Speaker: Stefan ANKER, Berlin, GER
Treatment optimization: Letting investigators decide vs. applying protocol defined algorithms (predefined, monitored, decision support systems)
Speaker: William T. ABRAHAM, Columbus, USA
What endpoint for securing approval (FDA, and in EU, beyond CE mark) and reimbursement
Debate led by regulatory agencies: Andrew FARB, FDA, USA - Ileana PIÑA, New York, USA
Intervention / Responsable scientifique
Thème
Documentation
Liens
Dans la même collection
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : New indications: Is hear…
DeliargyrisEfthymiosMODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Lunch Session 1 : Industry perspectiv…
WoehrleHolgerMODIGLIANI Lunch Debate Session 1 - Friday November 30, 2012 THE DEVICE THERAPY TRIALISTS WORKSHOP Chairpersons: Gaetano DE FERRARI, Pavia, ITA - Ileana PIÑA, New York, USA Webcast: Tariq AHMAD,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : New indications: Is hear…
PrasadKrishnaMODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : Different doses, differe…
VerheugtFreekMODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Lunch Session 1 : Non randomized and/…
PocockStuart J.MODIGLIANI Lunch Debate Session 1 - Friday November 30, 2012 THE DEVICE THERAPY TRIALISTS WORKSHOP Chairpersons: Gaetano DE FERRARI, Pavia, ITA - Ileana PIÑA, New York, USA Webcast: Tariq AHMAD,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : New indications: Is hear…
ZannadFaiezMODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : How to secure the optima…
GibsonMichaelMODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : Industry viewpoint (Joer…
KoeckJean-LouisMODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 2 : Well Established Methods…
KoenigWolfgangMODIGLIANI Workshop 2 - Friday November 30, 2012 : ATHEROSCLEROSIS IMAGING IN CLINICAL TRIALS Facilitating the discovery of effective therapies Chairpersons: Jagat NARULA, New York, USA - Ahmed
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Lunch Session 1 : Options of and alte…
AbrahamWilliam T.MODIGLIANI Lunch Debate Session 1 - Friday November 30, 2012 THE DEVICE THERAPY TRIALISTS WORKSHOP Chairpersons: Gaetano DE FERRARI, Pavia, ITA - Ileana PIÑA, New York, USA Webcast: Tariq AHMAD,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Workshop 1 : How to secure the optima…
GellerNancy L.MODIGLIANI Workshop 1 - Friday November 30, 2012 : THE THROMBOSIS TRIALISTS WORKSHOP DOSE AND TARGET PATIENT POPULATIONS ISSUES Chairpersons: Peter CLEMMENSEN, Copenhagen, DEN - George-Andrei DAN,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Debate Session 5 : Ultrafiltration fo…
RossiGian PaoloMODIGLIANI Debate Session 5 - Saturday December 1st, 2012 NOVEL DIURETIC STRATEGIES IN HEART FAILURE Chairpersons: Keld KJELDSEN, Copenhagen, DEN - Gian Paolo ROSSI, Padua, ITA Webcast: Patrick
Avec les mêmes intervenants et intervenantes
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Lunch Session 1 : Options of and alte…
AbrahamWilliam T.MODIGLIANI Lunch Debate Session 1 - Friday November 30, 2012 THE DEVICE THERAPY TRIALISTS WORKSHOP Chairpersons: Gaetano DE FERRARI, Pavia, ITA - Ileana PIÑA, New York, USA Webcast: Tariq AHMAD,
-
Cardiovascular Clinical Trialists (CVCT) Forum – Paris 2012 - Debate Session 5 : The Vaptans story …
AbrahamWilliam T.MODIGLIANI Debate Session 5 - Saturday December 1st, 2012 NOVEL DIURETIC STRATEGIES IN HEART FAILURE Chairpersons: Keld KJELDSEN, Copenhagen, DEN - Gian Paolo ROSSI, Padua, ITA Webcast: Patrick
Sur le même thème
-
La naissance de la médecine scientifique (par Pierre Corvol)
CorvolPierreMontenotJeanLa naissance de la médecine scientifique Dans La Maison Nuncingen (1837), Balzac met en scène une conversation entre quatre journalistes échauffés par un bon repas. L’un des commensaux, Émile
-
Prévention de l'accident vasculaire cérébral
Chaque année, en France, près de 125 000 cas d'AVC -- Accident Vasculaire Cérébral -- sont recensés. Avec les récidives, ce chiffre augmente de 25 % L' AVC représente la 1ère cause de handicap et
-
Du coté de chez...Michel Haïssaguerre
GloinecYvesPreNadègeInspiré du questionnaire de Proust, cet entretien plus intimiste engage une réflexion sur Michel Haïssaguerre en tant qu’homme et non plus en tant que cardiologue. Ce face-à-face permet de
-
Michel Haïssaguerre, entre rythmes et musicalité
GloinecYvesPreNadègeA l’occasion de la création de l’Institut Hospitalo-Universitaire LIRYC (Institut de Rythmologie et de Modélisation Cardiaque) dont il est à l’origine, le 2e volet de notre série Trip TIC
-
Cardio-vasculaire
GayBernardGossePhilippeDouardHervéSassoustGérardJournées Bordeaux Segalen 2013 - Formation Médicale Continue des Médecins Généralistes - Session Cardio-vasculaire
-
Syndrome d'aorte douloureuse
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,
-
Hypertension artérielle
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,
-
Myocardiopathies restrictives
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,
-
La fonction systolique
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,
-
Échographie de contraste
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,
-
Infarctus du myocarde
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,
-
Prothèses valvulaires
LafitteStéphaneCottarre-LafitteMarianneRéantPatriciaRoudautRaymondCe cours vidéo est tiré du cédérom Échocardiographie Doppler (éd. 2011), comportant plus de trente leçons (Écho-doppler transthoracique, Fonction systolique, Valvulopathies, Myocardiopathies,