Notice
A new preclinical model to test the response of breast cancer to genomically targeted therapy
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
Translational science : how to move from biology to clinical applications
The rapid progress in next generation DNA sequencing technology (NGS) means it is now possible, in principle, to obtain a complete list of the genetic abnormalities in a tumour. In practice, clinicians use NGS to obtain a much shorter list of "actionable" mutations that can be targeted with available drugs or used as prognostic markers. Unfortunately, the mere presence of an actionable mutation does not guarantee a response to the cognate drug, as shown by the SAFIR1 study (André et al 2014). To spare patients unnecessary exposure to experimental drugs, we need to prove that their tumours are addicted to the oncogenes we are trying to target. In practice, this means validating proposed treatments on patient-derived xenografts (PDX) before treating patients. This explains the resurgence in interest in PDXs, but the low take rate and slow growth rate of hormone receptor-positive (ER+) breast tumours make it difficult to obtain drug response data in a clinically meaningful timeframe, if at all. Injection of tumour cells into the mammary ducts is a new approach to xenografting developed by Dan Medina's lab to study ductal carcinoma in situ (DCIS). DCIS is notoriously difficult to graft, but Medina and colleagues reported a success rate of nearly 80% with ER+ DCIS. What this tells us is that the mammary ducts create a special microenvironment that provides ideal conditions for the survival and growth of ER+ DCIS cells. We have used intraductal injection to study preneoplastic and invasive breast cancer cells. By injecting genetically engineered preneoplastic human cells into the ducts through the nipple we can completely replace the murine luminal layer with a morphologically normal layer of human luminal cells, a technique we call "humanisation" of the mouse mammary ductal tree (Verbeke et al, submitted). We are now developing protocols to measure the therapeutic response of human patient-derived breast cancer xenografts, with the goal of providing feedback to clinicians within 8 weeks of taking biopsies. We see the combination of NGS and PDX in the neoadjuvant setting as a possible solution to the problem of identifying and killing lethal clones in heterogeneous breast tumours.
Cette présentation a été donnée dans le cadre du ScientificBRIO Day 2 organisé annuellement par le SIRIC BRIO et qui a pour but de réunirtous les acteurs du SIRIC BRIO et plus largement de la cancérologie à Bordeaux.
Thème
Dans la même collection
-
Bordeaux Cancer Initiative
DESSIS Damien
ELUSTONDO Frédéric
Translational science : how to move from biology to clinical applications Upon request of the University, the SIRIC was mandated to develop a transversal cancer axis at the University. The axis
-
Sociology contribution to understanding the (no)diffusion of a medical innovation
GORRY Philippe
Interface technology/clinical/SHS/public health : case study interventional radiology We will present results of an undergoing study on the socioeconomic processes of recognition in oncology of a
-
Expert panel discussions : why translational research matters
CAMERON David
En point d'orgue du Scientific BRIO Day, David Cameron, spécialiste reconnu en Oncologie, Professeur à l'Université d'Edimbourg, anime un débat sur la question de la recherche dite translationnelle,
-
-
-
-
-
-
Cost-effectiveness analysis for decision making, illustrated theory
BéNARD Antoine
Interface technology/clinical/SHS/public health : case study interventional radiology In a health care system with limited resources, each euro invested in a health care intervention is no longer
-
Translational cancer genomics and proteomics
SLEIJFER Stefan
Translational science : how to move from biology to clinical applications Evaluation of cell-free DNA (cfDNA) is a very attractive tool to serve as “liquid biopsy” to define and establish
-
BRIO's facts and figures, structuration projects : Axe Cancer, translational research centerUpdate …
SOUBEYRAN Pierre
Update the implementation of BRIO Brio organisation is now fully up and running, and more than 100 research projects are currently active into the 6 integrated research programmes. In addition,
Sur le même thème
-
Place du brevet dans la recherche d'un laboratoire
DUTREIX Marie
GIRARD Pierre-Marie
Interview : la place du brevet dans la recherche d'un laboratoire par Marie Dutreix, directrice de recherche à l'institut Curie.
-
Influence des comportements de santé sur le jugement des femmes consommatrices et atteintes de canc…
AURIOL Camille
Les doctorants des universités de Toulouse (UT1C/UT2J/UT3) et de l'Université de Québec à Montréal (UQAM) ont organisé un séminaire de présentation et d'échanges autour de leurs travaux de recherche,
-
[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)
NICAISE Sarah
MAYèRE Anne
TERRAL Philippe
GABORIT Emilie
DEFOSSEZ Adrien
BARTHE Jean-François
Symposium International FAB.ISS - Ce symposium proposé en 2020 en visioconférence s'intéresse à la fabrique des inégalités sociales de santé, du point de vue des disciplines de sciences humaines et
-
Explorer les impensés de dispositifs de télé-surveillance sur les patient.e.s et leurs modes de vie
MAYèRE Anne
La conférence du Pr. Anne Mayère (CERTOP, IFERISS, Université de Toulouse, France) est organisée dans le cadre du séminaire télésanté de la Chaire de recherche en francophonie internationale sur les
-
Les jeudis du Grhapes : « Approches sociologiques de l'inclusion », Claude-Julie Bourque, sociolog…
« Approches sociologiques de l'inclusion » Guérir, et puis après... ? Claude-Julie Bourque, sociologue, professeure-chercheure adjointe au département de pédiatrie de la Faculté de médecine de
-
SFjRO Rouen 2019 - Radiosensibilité et apoptose lymphocytaire
Radiosensibilité et apoptose lymphocytaire
-
SFjRO Rouen 2019 - Applications cliniques de la protons thérapie
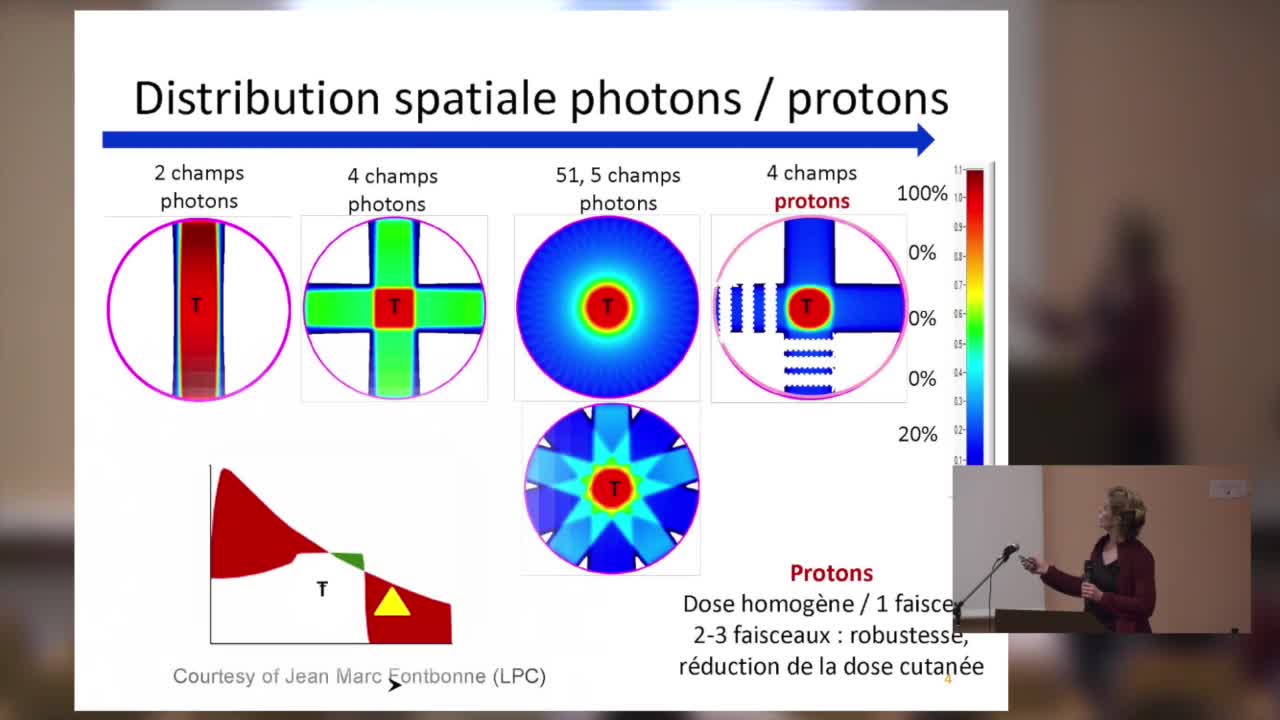
Applications cliniques de la protons thérapie
-
SFjRO Rouen 2019 - Association therapie systémique et radiothérapie
Association therapie systémique et radiothérapie
-
SFjRO Rouen 2019 - Bases physiques et biologie de la proton thérapie
Bases physiques et biologie de la proton thérapie
-
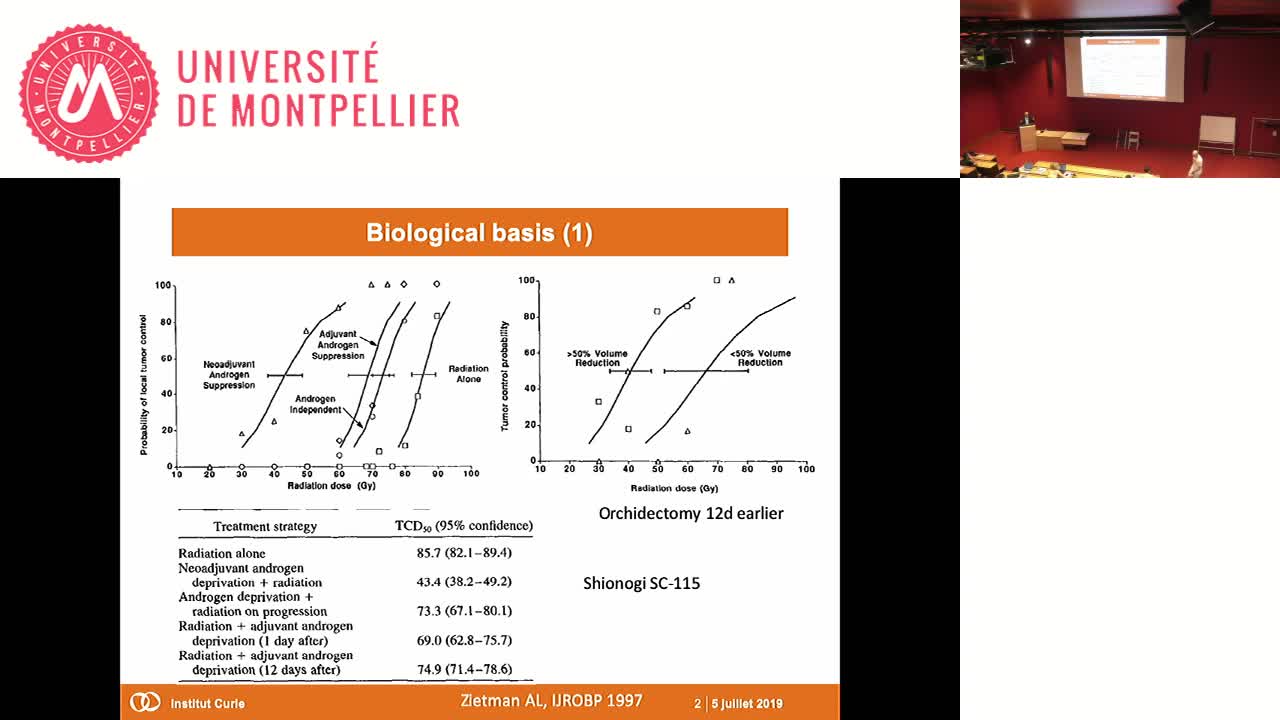
SFjRO Montpellier 2019 - Radio-hormonothérapie
SFjRO Montpellier 2019 - Radio-hormonothérapie
-
-
SFjRO Montpellier 2019 - Hypofractionnement
SFjRO Montpellier 2019 - Hypofractionnement















![[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)](https://vod.canal-u.tv/videos/media/images/iferiss/.fab.iss.session.5.la.fabrique.des.inegalites.sociales.de.sante.et.cancer.s._59509/7.png)