Chapitres
- >> Frédéric Elustondo01'38"
- >> Harold Wodrich15'39"
- >> Benjamin Faustin13'07"
- >> Frédéric Saltel17'10"
- >> Laurent Cognet15'13"
Notice
Flash talk - Call for proposal FAC 2014
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
Personalized breast cancer therapy based on viral functional assays toscore pathway activity
HaraldWodrich, University of Bordeaux
State-of-the-art mass sequencingtechnology has paved the road to identify the genetic make-up of tumors on apatient-by-patient basis, allowing targeted and personalized treatment. Whilethis worked in specific cases it remains difficult to translate genetic changesinto causative cancer pathway alterations. For example, in luminal A breast tumours PIK3CA mutations that activatethe kinase do not correlate with phosphorylation of downstream targets orresponse to PI3K inhibitors. An alternative/complementing approach is todirectly test pathway integrity in primary tumor samples. We propose to useadenovirus infection of primary breast cancer samples as such a test.Adenoviruses modulate and depend on several of the pathways frequently mutatedin breast cancer to deliver their genomes into the nucleus. This property isentirely encoded in the virion and does not require viral gene expression. Hence weattempt to score pathways by comparing marker gene expression from wildtype virionswith mutant virions developed in our group that are sensitive to defects in thePI3K/AKT/mTOR/SGK1 pathway and subject to autophagic degradation. Since these events occur so early, they are ideallyplaced to form the basis of rapid functional assays for oncogenic pathwayactivation.
Activation of human gamma-delta T cells by cancer metabolism
BenjaminFaustin, University of Bordeaux
Compelling clinical data frompatients treated with immunomodulatory agents indicate that harnessing thepower of the immune system can improve long-term cancer containment and extendoverall survival. Immunotherapies potentiate or reactivate ongoing, inefficientantitumor immune responses and break tumor tolerance, which is one of the majorstrategies used by cancer cells to escape immune recognition. Human γδ T cells contribute to lymphoidstress surveillance against tumors by directly recognizing cancer cells throughunknown mechanisms. This year, we have shown that several types of cancer cellsexpress caspase-1 inflammasomes and release IL-18 to promote T cellcytotoxicity and IFNγproduction. Moreover, we report that triggering the AMP-activated proteinkinase (AMPK)-dependent metabolic reprogramming of cancer cells induces theupregulation of a broad immune signature comprised of T-Cell Receptor antigens,co-stimulatory molecules, adhesion molecule, and the secretion of solublemolecules. These signals merge to enhance γδ T cell response and potentially others immunecells. Hence, AMPK activity in cancer cells improves immune detection by tissueγδ T cells withimplications for cancer immunotherapy. This research is applied to colorectaland lung cancers (which are sites of γδ T cell homing).
Discoidin domain receptors (DDR) involvement in melanoma progression andinvasion
FrédéricSaltel, University of Bordeaux
Background:Cutaneous melanoma is an aggressive cancer that forms from melanocytes. Duringmelanoma progression, cells will invade the dermis which is rich in type Icollagen. Linear invadosomes are invasive structures induced by fibrillar typeI collagen, able to degrade the matrix. The discoidin domain receptor 1 (DDR1)was shown to be necessary for linear invadosomes formation and functionality.DDR1 and DDR2 are overexpressed or mutated in various tumors, mediating cellproliferation and/or invasion. DDR1 is known to mediate cell migration andinvasion in non-small cell lung carcinoma and DDR2 has been shown to facilitatebreast cancer cells migration and metastasis. However there is no solidevidence of DDRs involvement in melanoma. As the melanoma cells will be incontact with type I collagen
fibers duringinvasion into the dermis, our hypothesis is that if melanoma express DDRs,linear invadosomes may be relevant in melanoma progression.
Results: Ourpreliminary data obtained on human melanoma tissue samples demonstrate thatDDR1 and DDR2 are co-overexpressed in this cancer. In vitro, a comparisonbetween primary human melanocytes and a melanoma cell lines shows that DDRs areoverexpressed in A375. Moreover, DDR1 and DDR2 colocalize along collagenfibers. Furthermore, melanoma cell lines are able to form more linearinvadosomes than primary melanocytes. These results prompted us to study theinvolvement of DDR1 and DDR2 in a same cellular model, addressing thecontribution of each member in melanoma progression and linear invadosomesformation. Using an ex vivo model of reconstructed epidermis, we will study theimpact of overexpression or depletion of DDR1 and/or DDR2 on cell invasion. Inparallel, we want to highlight the existence of linear invadosomes in vivousing correlative microscopy. Finally, we want to determine DDRs as potential therapeutictargets to block tumor invasion in melanoma.
Quantitative optical nanoscopy of infrared nano-markers : toward colon cancer tissue microarrays characterization
LaurentCognet, University of Bordeaux
The main objective of this project isthe simultaneous development, implementation and tests of a new imagingmodality, Photothermal Imaging (PhI) that can detect single absorbingnanolabels. Our aim is to supplement IHC and IF in terms of sensitivity, andreproducibility and transfer this approach to the medical world in order to aidcancer diagnosis and to guide therapeutic choices.
In this presentation, I will show that thefirst tests performed on human cancer samples from the tumor bank of Bergoniéindicate that PhI should be operating with near infrared nano-labels. I willthen present our current efforts to produce and characterize such near infrarednano-labels. Those are based on extremely small gold nanorods and ultra-shortnanotubes. Future experiments including nanoparticle bio-functionalization andimaging microarrays of colon tumors will then be discussed.
Ces présentations ont été données dans le cadre du BRIO SIRICscientific day 3 organisé annuellement par le SIRIC BRIO et qui a pour but deréunir tous les acteurs du SIRIC BRIO et plus largement de la cancérologie àBordeaux.
Intervention / Responsable scientifique
Thème
Dans la même collection
-
-
Experts panel discussion: What opportunities and best practices for the use of clinical material in…
CameronDavidEn point d'orgue du Scientific BRIO Day, David Cameron, spécialiste reconnu en Oncologie, Professeur à l'Université d'Edimbourg, anime un débat sur la question des opportinités et bonnes pratiques
-
A combined laser microdissection and mass spectrometry method for proteomic analysis of tissue sect…
RaymondAnne-AurélieClinical and biological samples : opportunities and challenges for translational research A challenge in oncology is to better define the tumor of the patient to study the pathology and of course
-
A new weapon against the tobacco industry : class action lawsuit using a novel epidemiologic param…
SiemiatyckiJackEpidemiology and public health Lawsuits against the tobacco industry, if successful, have the potential to compensate victims of smoking and to diminish the capacity of the tobacco industry to
-
BRIO strategic challenges and the implementation of a translational culture in Bordeaux
SoubeyranPierreAbout BRIO Siric challenges and actions BRIO’s strategic objective is to strengthen Bordeaux’s leadership in Cancer Research through the creation of a dynamic cancer research community, the
-
Social inequalities impacts of care management and survival in patients with non-hodgkin lymphomas
Le Guyader-PeyrouSandraEpidemiology and public health The principal aim of this thesis will describe the care pathway of diffuse large B cell lymphoma (DBCL) and follicular lymphoma (FL) in the general population and
-
BRIO supporting action to integrated cancer research in Bordeaux
MeoniPaoloAbout BRIO Siric challenges and actions The success of BRIO’s approach and the realization of its potential impact on the translational research community in Bordeaux rely both on the efficient
-
-
How studying hematological malignancies subtypes may inform epidemiologic and clinical research
MonnereauAlainEpidemiology and public health The aim of the talk entitled "How studying hematological malignancies (HM) subtypes may inform epidemiologic and clinical research" is to bring to the audience
-
Sarcoma Database: a unique tool for research on mesenchymal tumors
CoindreJean-MichelClinical biology session The main sarcoma databases are the Conticabase (https://conticabase.sarcomabcb.org) and the ConticaGist (https://conticagist.sarcomabcb.org) with 18000 patients and which
-
Modelling prognostic capabilities of tumor size : application to colorectal cancer
RondeauVirginieEpidemiology and public health In oncology, the international WHO and RECIST criteria have allowed the standardization of tumor response evaluation in order to identify the time of disease
Avec les mêmes intervenants et intervenantes
-
Bordeaux Cancer Initiative
DessisDamienElustondoFrédéricTranslational science : how to move from biology to clinical applications Upon request of the University, the SIRIC was mandated to develop a transversal cancer axis at the University. The axis
Sur le même thème
-
Utiliser la lumière pour comprendre la matière
ChapronDavidDavid Chapron, enseignant-chercheur du LMOPS (Laboratoire Matériaux optiques, photonique et systèmes), présente la technique de spectroscopie Raman dont il est l'un des spécialistes français.
-
02. Faire de son mieux (Avec Nathalie Vallet-Renart)
Vallet-RenartNathaliePodcast animé par Romain Poncet, ingénieur de recherche en sociologie.
-
01. Le soin du détail (Avec Emilie Chanel)
PoncetRomainChanelEmilieCe podcast est proposé par la Chaire Valeurs du Soin, animé par Romain Poncet, ingénieur de recherche en sociologie.
-
Place du brevet dans la recherche d'un laboratoire
DutreixMarieGirardPierre-MarieInterview : la place du brevet dans la recherche d'un laboratoire par Marie Dutreix, directrice de recherche à l'institut Curie.
-
Influence des comportements de santé sur le jugement des femmes consommatrices et atteintes de canc…
AuriolCamilleLes doctorants des universités de Toulouse (UT1C/UT2J/UT3) et de l'Université de Québec à Montréal (UQAM) ont organisé un séminaire de présentation et d'échanges autour de leurs travaux de recherche,
-
[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)
NicaiseSarahMayèreAnneTerralPhilippeGaboritEmilieDefossezAdrienBartheJean-FrançoisSymposium International FAB.ISS - Ce symposium proposé en 2020 en visioconférence s'intéresse à la fabrique des inégalités sociales de santé, du point de vue des disciplines de sciences humaines et
-
Explorer les impensés de dispositifs de télé-surveillance sur les patient.e.s et leurs modes de vie
MayèreAnneLa conférence du Pr. Anne Mayère (CERTOP, IFERISS, Université de Toulouse, France) est organisée dans le cadre du séminaire télésanté de la Chaire de recherche en francophonie internationale sur les
-
Les jeudis du Grhapes : « Approches sociologiques de l'inclusion », Claude-Julie Bourque, sociolog…
« Approches sociologiques de l'inclusion » Guérir, et puis après... ? Claude-Julie Bourque, sociologue, professeure-chercheure adjointe au département de pédiatrie de la Faculté de médecine de
-
SFjRO Rouen 2019 - Association therapie systémique et radiothérapie
Association therapie systémique et radiothérapie
-
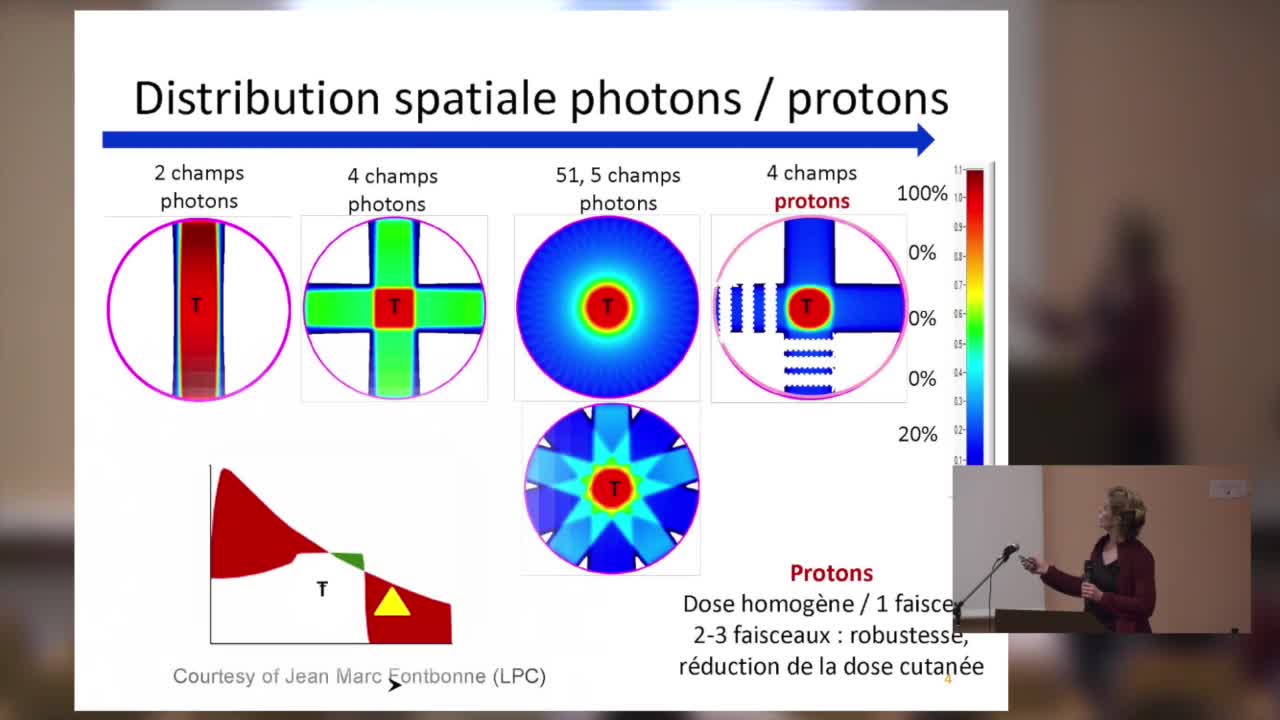
SFjRO Rouen 2019 - Bases physiques et biologie de la proton thérapie
Bases physiques et biologie de la proton thérapie
-
SFjRO Rouen 2019 - Radiosensibilité et apoptose lymphocytaire
Radiosensibilité et apoptose lymphocytaire
-
SFjRO Rouen 2019 - Applications cliniques de la protons thérapie
Applications cliniques de la protons thérapie


















![[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)](https://vod.canal-u.tv/videos/media/images/iferiss/.fab.iss.session.5.la.fabrique.des.inegalites.sociales.de.sante.et.cancer.s._59509/7.png)