Notice
Life of a sample
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
Clinicaland biological samples : opportunities and challenges for translationalresearch
The first purpose of this talk is to provide tonnon clinical researchers some main technical, ethical and reglementary issuesfor their access to biological samples and especially tumoral tissues which arestored in pathology departments or tumorbanks. The second one is to explain toscientists that their project will not be successful if the sample collectionis not associated with relevant clinical and biopathological annotations
Finally, financial and ethical problems will bediscussed as cancer biobanks funding becomes problematic and there is also anurgent need to refine their policy considering the most frequent tumor subtypeswhich may be available in several biobanks with redundancy and the access ofresearchers to routine diagnostic samples or rare tumor subtypes which are notprimarily stored for research but that can be qualified for research by anEthical Committee.
Samples collected in daily practice are ofdiverse origins and subtypes: freshtissues, cell suspension, fixed biopsies, surgical specimens. All of them maybe altered by adverse preanalytical factors such as cold ischemia, delayedfixation or freezing times which are not always known parameters. Therefore,research collection can be set up with knowledge of these parameters andalternative procedures such as RNA later prior to freezing. Accreditation ofpathology department will also make possible to limit preanalytical artefactswhich are critical according to the tissue subtype and technique used foranalysis with robust and more sensitive ones.
The research collection needs to be associatedwith basic clinical and pathological parameters but all of them are onlyavailable when a clinical database with evolutive features is designed andmaintained. Despite the efforts of these clinical databases, the life of thesample needs to be managed independently at the biopathology level. In factthere are two different situations: oneis when the cancer biobank and the biopathology department directly involved inthe research project. Another seen at the Bordeaux University Hospital is whenmore than 10 research collection are setup in the context of regional, nationalor international project with centralized management and funding of theclinical database as well as of the scientific advisory board for access to thesamples. Despite the presence of suchclinical database, the scientific project cannot be conducted without aspecific reanalysis of biopathological data owing to the diversity of tumourhistological and genetic subtypes, especially when changes in theclassification of the tumor subtypes (lung, liver, lymphoma for example) haveoccurred. Both clinicians and scientists are not aware that their project willnot only need funding for access to tissue but that they will need to involvean expert biopathologist in their project.
Whatever the tumor bank subtype, the processfor storing, management and sample access are regulated through certifiedprocedure NFS 96900 in France. Finally, the access to sample is restricted byethical and regulatory procedures for which the tumor bank is not in charge buthas together with the researcher to cope with.
Cette présentation a été donnée dans le cadre du BRIO SIRICscientific day 3 organisé annuellement par le SIRIC BRIO et qui a pour but deréunir tous les acteurs du SIRIC BRIO et plus largement de la cancérologie àBordeaux.
Thème
Dans la même collection
-
Flash talk - Call for proposal FAC 2014
ElustondoFrédéricWodrichHaraldFaustinBenjaminSaltelFrédéricCognetLaurentPersonalized breast cancer therapy based on viral functional assays to score pathway activity Harald Wodrich, University of Bordeaux State-of-the-art mass sequencing technology has paved the road
-
-
Experts panel discussion: What opportunities and best practices for the use of clinical material in…
CameronDavidEn point d'orgue du Scientific BRIO Day, David Cameron, spécialiste reconnu en Oncologie, Professeur à l'Université d'Edimbourg, anime un débat sur la question des opportinités et bonnes pratiques
-
A new weapon against the tobacco industry : class action lawsuit using a novel epidemiologic param…
SiemiatyckiJackEpidemiology and public health Lawsuits against the tobacco industry, if successful, have the potential to compensate victims of smoking and to diminish the capacity of the tobacco industry to
-
A combined laser microdissection and mass spectrometry method for proteomic analysis of tissue sect…
RaymondAnne-AurélieClinical and biological samples : opportunities and challenges for translational research A challenge in oncology is to better define the tumor of the patient to study the pathology and of course
-
Social inequalities impacts of care management and survival in patients with non-hodgkin lymphomas
Le Guyader-PeyrouSandraEpidemiology and public health The principal aim of this thesis will describe the care pathway of diffuse large B cell lymphoma (DBCL) and follicular lymphoma (FL) in the general population and
-
BRIO strategic challenges and the implementation of a translational culture in Bordeaux
SoubeyranPierreAbout BRIO Siric challenges and actions BRIO’s strategic objective is to strengthen Bordeaux’s leadership in Cancer Research through the creation of a dynamic cancer research community, the
-
How studying hematological malignancies subtypes may inform epidemiologic and clinical research
MonnereauAlainEpidemiology and public health The aim of the talk entitled "How studying hematological malignancies (HM) subtypes may inform epidemiologic and clinical research" is to bring to the audience
-
BRIO supporting action to integrated cancer research in Bordeaux
MeoniPaoloAbout BRIO Siric challenges and actions The success of BRIO’s approach and the realization of its potential impact on the translational research community in Bordeaux rely both on the efficient
-
Modelling prognostic capabilities of tumor size : application to colorectal cancer
RondeauVirginieEpidemiology and public health In oncology, the international WHO and RECIST criteria have allowed the standardization of tumor response evaluation in order to identify the time of disease
-
Sarcoma Database: a unique tool for research on mesenchymal tumors
CoindreJean-MichelClinical biology session The main sarcoma databases are the Conticabase (https://conticabase.sarcomabcb.org) and the ConticaGist (https://conticagist.sarcomabcb.org) with 18000 patients and which
Sur le même thème
-
01. Le soin du détail (Avec Emilie Chanel)
Ce podcast est proposé par la Chaire Valeurs du Soin, animé par Romain Poncet, ingénieur de recherche en sociologie.
-
02. Faire de son mieux (Avec Nathalie Vallet-Renart)
Vallet-RenartNathaliePodcast animé par Romain Poncet, ingénieur de recherche en sociologie.
-
Place du brevet dans la recherche d'un laboratoire
DutreixMarieGirardPierre-MarieInterview : la place du brevet dans la recherche d'un laboratoire par Marie Dutreix, directrice de recherche à l'institut Curie.
-
Influence des comportements de santé sur le jugement des femmes consommatrices et atteintes de canc…
AuriolCamilleLes doctorants des universités de Toulouse (UT1C/UT2J/UT3) et de l'Université de Québec à Montréal (UQAM) ont organisé un séminaire de présentation et d'échanges autour de leurs travaux de recherche,
-
[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)
NicaiseSarahMayèreAnneTerralPhilippeGaboritEmilieDefossezAdrienBartheJean-FrançoisSymposium International FAB.ISS - Ce symposium proposé en 2020 en visioconférence s'intéresse à la fabrique des inégalités sociales de santé, du point de vue des disciplines de sciences humaines et
-
Explorer les impensés de dispositifs de télé-surveillance sur les patient.e.s et leurs modes de vie
MayèreAnneLa conférence du Pr. Anne Mayère (CERTOP, IFERISS, Université de Toulouse, France) est organisée dans le cadre du séminaire télésanté de la Chaire de recherche en francophonie internationale sur les
-
Les jeudis du Grhapes : « Approches sociologiques de l'inclusion », Claude-Julie Bourque, sociolog…
« Approches sociologiques de l'inclusion » Guérir, et puis après... ? Claude-Julie Bourque, sociologue, professeure-chercheure adjointe au département de pédiatrie de la Faculté de médecine de
-
SFjRO Rouen 2019 - Radiosensibilité et apoptose lymphocytaire
Radiosensibilité et apoptose lymphocytaire
-
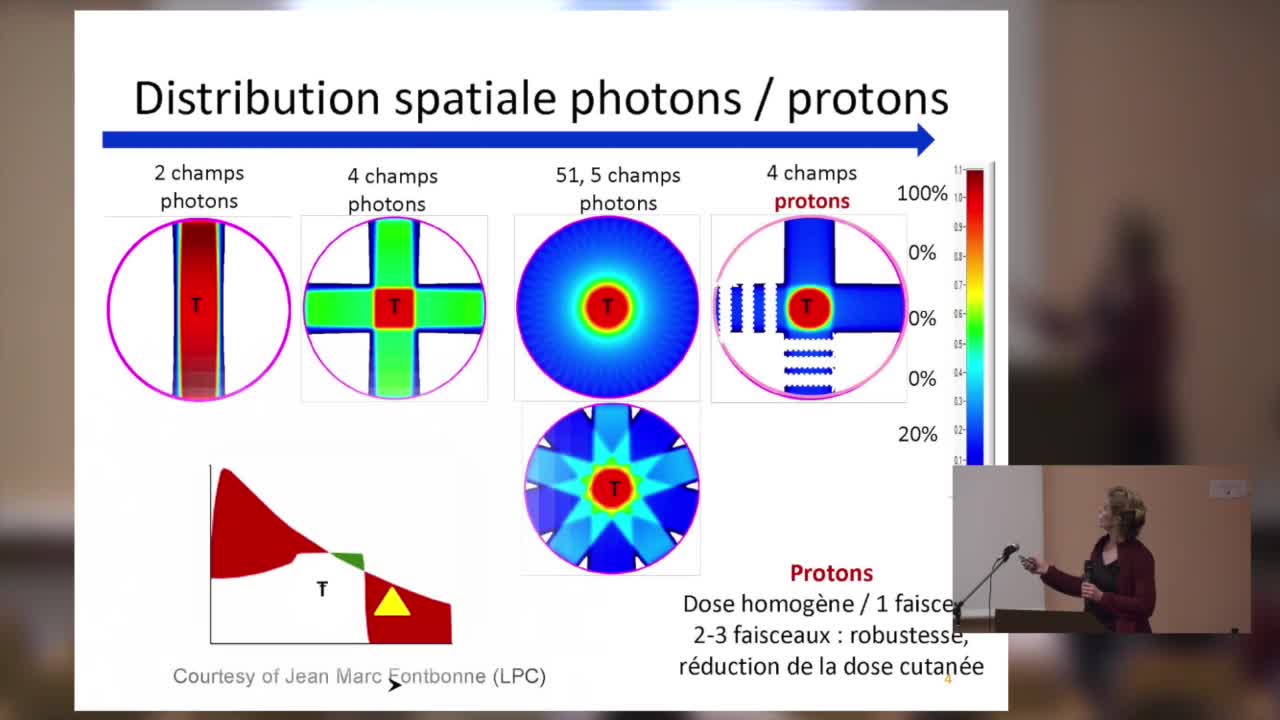
SFjRO Rouen 2019 - Applications cliniques de la protons thérapie
Applications cliniques de la protons thérapie
-
SFjRO Rouen 2019 - Association therapie systémique et radiothérapie
Association therapie systémique et radiothérapie
-
SFjRO Rouen 2019 - Bases physiques et biologie de la proton thérapie
Bases physiques et biologie de la proton thérapie
-
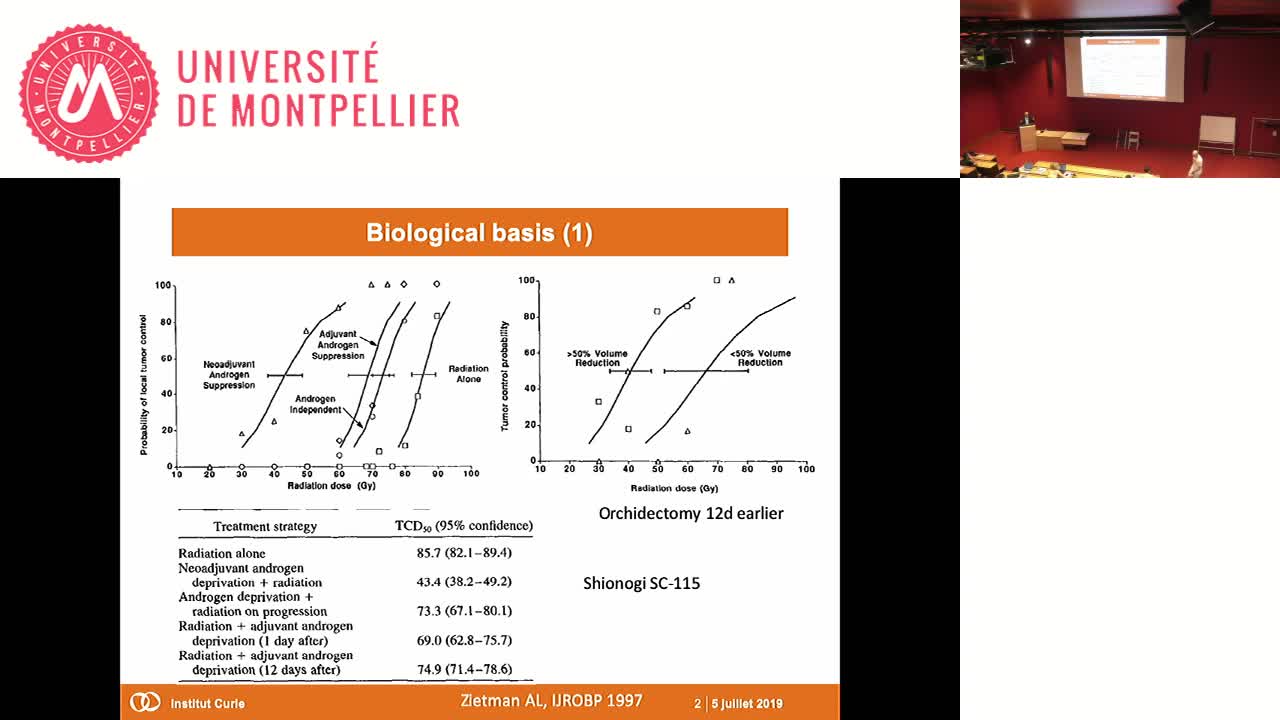
SFjRO Montpellier 2019 - Radio-hormonothérapie
SFjRO Montpellier 2019 - Radio-hormonothérapie
















![[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)](https://vod.canal-u.tv/videos/media/images/iferiss/.fab.iss.session.5.la.fabrique.des.inegalites.sociales.de.sante.et.cancer.s._59509/7.png)