Notice
Modelling prognostic capabilities of tumor size : application to colorectal cancer
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
Epidemiologyand public health
In oncology, the international WHO and RECISTcriteria have allowed the standardization of tumor response evaluation in orderto identify the time of disease progression. These semi-quantitativemeasurements are often used as endpoints in phase II and phase III trials tostudy the efficacy of new therapies. However, through categorization of thecontinuous tumor size, information can be lost and they can be challenged byrecently developed methods of modeling biomarkers in a longitudinal way. Thus,it is of interest to compare the predictive ability of cancer progressionsbased on categorical criteria and quantitative measures of tumor size(left-censored due to detection limit problems) and/or appearance of new lesionson overall survival. We propose a joint model for a simultaneous analysis ofthree types of data: longitudinal marker, recurrent events and a terminalevent. A simulation study is performed and shows that the proposed trivariatemodel is appropriate for the practical use. We suggest statistical tools thatevaluate predictive accuracy for joint models to compare our model to modelsbased on categorical criteria and their components. We apply the model to arandomized phase III clinical trial of metastatic colorectal cancer, conductedby the Fédération Francophone de Cancérologie Digestive (FFCD 2000-05 trial),which assigned 410 patients to two therapeutic strategies with multiplesuccessive chemotherapy regimens.
Cette présentation a été donnée dans le cadre du BRIO SIRICscientific day 3 organisé annuellement par le SIRIC BRIO et qui a pour but deréunir tous les acteurs du SIRIC BRIO et plus largement de la cancérologie àBordeaux.
Thème
Dans la même collection
-
Experts panel discussion: What opportunities and best practices for the use of clinical material in…
CAMERON David
En point d'orgue du Scientific BRIO Day, David Cameron, spécialiste reconnu en Oncologie, Professeur à l'Université d'Edimbourg, anime un débat sur la question des opportinités et bonnes pratiques
-
A new weapon against the tobacco industry : class action lawsuit using a novel epidemiologic param…
SIEMIATYCKI Jack
Epidemiology and public health Lawsuits against the tobacco industry, if successful, have the potential to compensate victims of smoking and to diminish the capacity of the tobacco industry to
-
A combined laser microdissection and mass spectrometry method for proteomic analysis of tissue sect…
RAYMOND Anne-Aurélie
Clinical and biological samples : opportunities and challenges for translational research A challenge in oncology is to better define the tumor of the patient to study the pathology and of course
-
Flash talk - Call for proposal FAC 2014
ELUSTONDO Frédéric
WODRICH Harald
FAUSTIN Benjamin
SALTEL Frédéric
COGNET Laurent
Personalized breast cancer therapy based on viral functional assays to score pathway activity Harald Wodrich, University of Bordeaux State-of-the-art mass sequencing technology has paved the road
-
-
How studying hematological malignancies subtypes may inform epidemiologic and clinical research
MONNEREAU Alain
Epidemiology and public health The aim of the talk entitled "How studying hematological malignancies (HM) subtypes may inform epidemiologic and clinical research" is to bring to the audience
-
BRIO supporting action to integrated cancer research in Bordeaux
MEONI Paolo
About BRIO Siric challenges and actions The success of BRIO’s approach and the realization of its potential impact on the translational research community in Bordeaux rely both on the efficient
-
-
Sarcoma Database: a unique tool for research on mesenchymal tumors
COINDRE Jean-Michel
Clinical biology session The main sarcoma databases are the Conticabase (https://conticabase.sarcomabcb.org) and the ConticaGist (https://conticagist.sarcomabcb.org) with 18000 patients and which
-
Social inequalities impacts of care management and survival in patients with non-hodgkin lymphomas
LE GUYADER-PEYROU Sandra
Epidemiology and public health The principal aim of this thesis will describe the care pathway of diffuse large B cell lymphoma (DBCL) and follicular lymphoma (FL) in the general population and
-
BRIO strategic challenges and the implementation of a translational culture in Bordeaux
SOUBEYRAN Pierre
About BRIO Siric challenges and actions BRIO’s strategic objective is to strengthen Bordeaux’s leadership in Cancer Research through the creation of a dynamic cancer research community, the
Sur le même thème
-
Place du brevet dans la recherche d'un laboratoire
DUTREIX Marie
GIRARD Pierre-Marie
Interview : la place du brevet dans la recherche d'un laboratoire par Marie Dutreix, directrice de recherche à l'institut Curie.
-
Influence des comportements de santé sur le jugement des femmes consommatrices et atteintes de canc…
AURIOL Camille
Les doctorants des universités de Toulouse (UT1C/UT2J/UT3) et de l'Université de Québec à Montréal (UQAM) ont organisé un séminaire de présentation et d'échanges autour de leurs travaux de recherche,
-
[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)
NICAISE Sarah
MAYèRE Anne
TERRAL Philippe
GABORIT Emilie
DEFOSSEZ Adrien
BARTHE Jean-François
Symposium International FAB.ISS - Ce symposium proposé en 2020 en visioconférence s'intéresse à la fabrique des inégalités sociales de santé, du point de vue des disciplines de sciences humaines et
-
Explorer les impensés de dispositifs de télé-surveillance sur les patient.e.s et leurs modes de vie
MAYèRE Anne
La conférence du Pr. Anne Mayère (CERTOP, IFERISS, Université de Toulouse, France) est organisée dans le cadre du séminaire télésanté de la Chaire de recherche en francophonie internationale sur les
-
Les jeudis du Grhapes : « Approches sociologiques de l'inclusion », Claude-Julie Bourque, sociolog…
« Approches sociologiques de l'inclusion » Guérir, et puis après... ? Claude-Julie Bourque, sociologue, professeure-chercheure adjointe au département de pédiatrie de la Faculté de médecine de
-
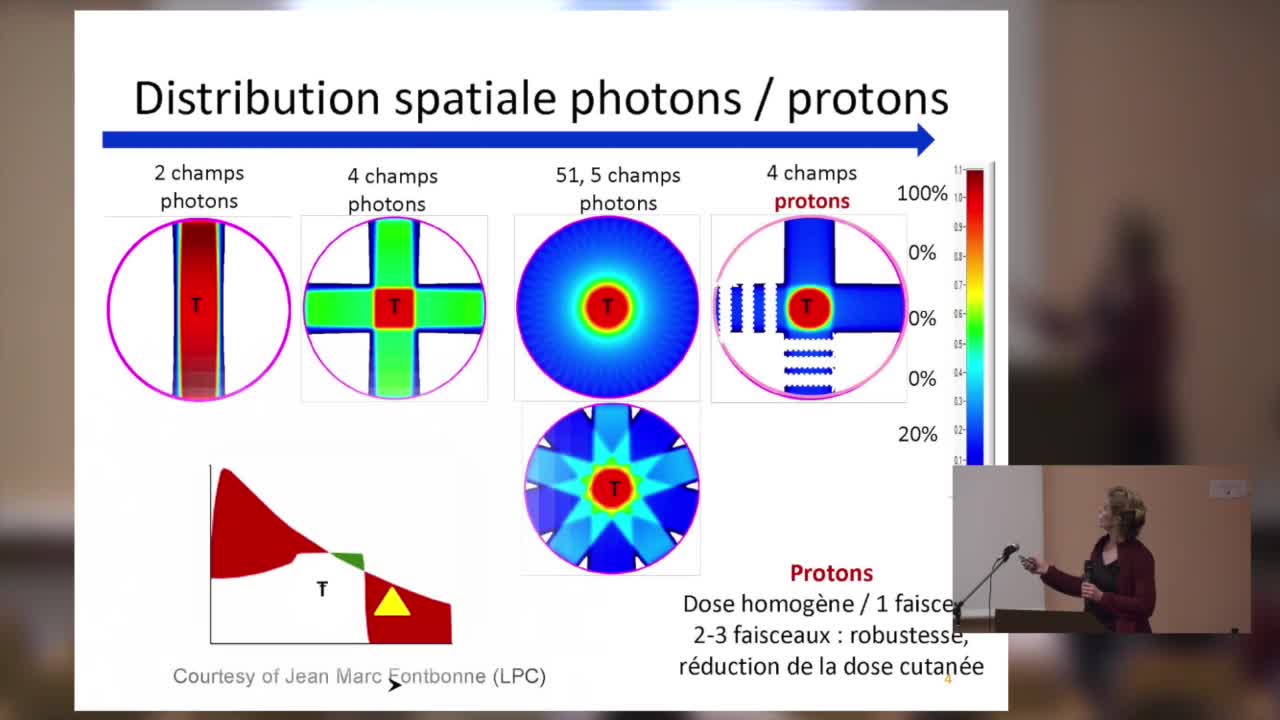
SFjRO Rouen 2019 - Bases physiques et biologie de la proton thérapie
Bases physiques et biologie de la proton thérapie
-
SFjRO Rouen 2019 - Radiosensibilité et apoptose lymphocytaire
Radiosensibilité et apoptose lymphocytaire
-
SFjRO Rouen 2019 - Applications cliniques de la protons thérapie
Applications cliniques de la protons thérapie
-
SFjRO Rouen 2019 - Association therapie systémique et radiothérapie
Association therapie systémique et radiothérapie
-
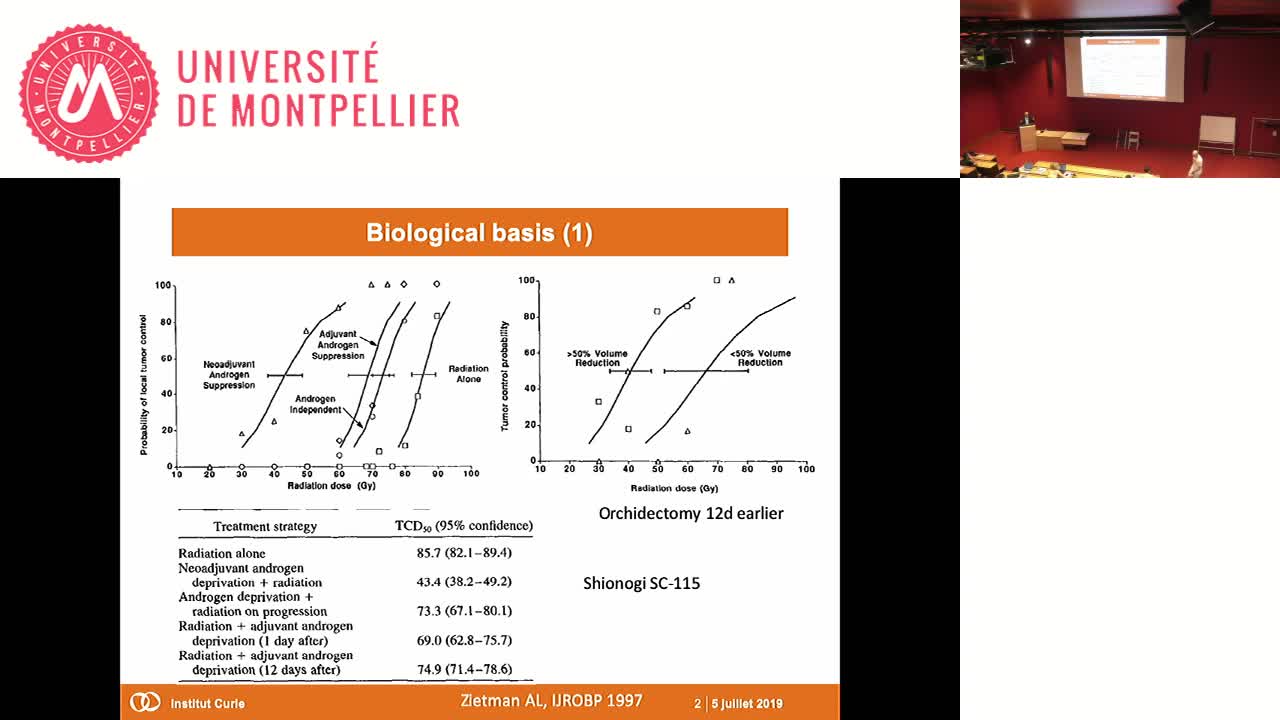
SFjRO Montpellier 2019 - Radio-hormonothérapie
SFjRO Montpellier 2019 - Radio-hormonothérapie
-
SFjRO Montpellier 2019 - Hypofractionnement
SFjRO Montpellier 2019 - Hypofractionnement
-
SFJRO Montpellier 2019 - Cancer prostate métastatique hormonosensible
SFJRO Montpellier 2019 - Cancer prostate métastatique hormonosensible















![[FAB.ISS] Session #5 La fabrique des inégalités sociales de santé et cancer(s)](https://vod.canal-u.tv/videos/media/images/iferiss/.fab.iss.session.5.la.fabrique.des.inegalites.sociales.de.sante.et.cancer.s._59509/7.png)