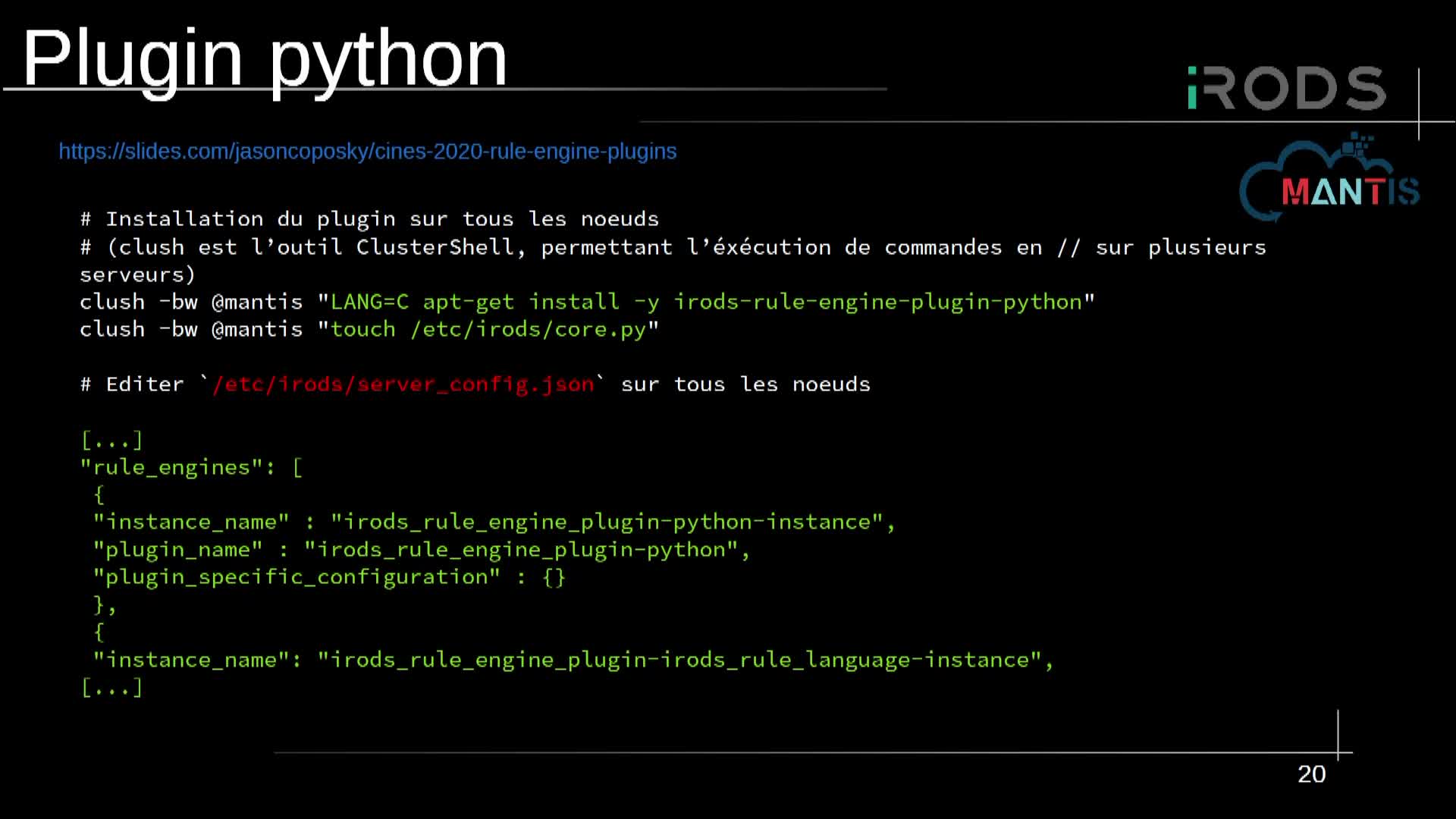
Notice
Mcl-1 conformational landscape exploration
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
Protein-protein interactions are attractive targets because they control numerous cellular processes. In oncology, apoptosis regulating Bcl-2 family proteins are of particular interest. Bcl-2 proteins are crucial regulators of the intrinsic mitochondrial pathway of apoptosis and comprise both pro-apoptotic and anti-apoptotic proteins.1 Apoptotic cell death is controlled via PPIs between the anti-apoptotic proteins hydrophobic groove and the pro-apoptotic proteins BH3 domain. Mcl-1 (an anti-apoptotic Bcl-2 member) is a key regulator of cancer cell survival and a known resistance factor to Bcl-2/Bcl-xL pharmacological inhibitors making it an attractive therapeutic target. The ability of Mcl-1 to bind different partners is possibly linked to the large plasticity of its binding groove, with four hydrophobic pockets, and hence to a wide conformational space accessible to this protein. We have developed recently in our laboratory a new family of original ligands able to bind to Mcl-1.2
In this study, we explore Mcl-1 conformational space on the basis of the crystallographic data available within the Protein data-bank (PDB), but also by using data harvested by conventional all-atom molecular dynamics simulations. For that purpose, methods to explore the essential dynamics such as principal component analysis (PCA)3, all-atom normal modes (ENM) are used to analyze this data. We successfully captured a breathing motion from both data-sets and we hypothesize that this breathing motion is a major structural feature that leads Mcl-1 to adapt to its binding partners.
Thème
Documentation
Dans la même collection
-
-
-
-
-
-
-
-
Recherche Data Gouv : un écosystème au service du partage et de l'ouverture des données de recherche
Un écosystème au service du partage et de l’ouverture des données de recherche
-
-
-
Refroidissement par immersion de serveurs : premiers retours opérationnels
Le refroidissement par immersion n'est pas une nouveauté, de l'IT en général au HPC en particulier. Il y a 35 ans, les Cray-2 et successeurs avaient leur assemblage de tours immergées dans un fluide
-