Notice
Dr Frédéric Marin - Mollusk shell matrices: unexpected functions in biomineralization
- document 1 document 2 document 3
- niveau 1 niveau 2 niveau 3
Descriptif
To construct their skeletons, all metazoans secrete a complex array ofmacromolecules that are supposed to display key-functions in biomineralization,such as crystal nucleation and crystal growth orientation. These macromolecules- generally less than 1% of the skeletal weight - are occluded during skeletalgrowth and can be retrieved and analyzed by dissolving the mineral phase. Theycomprise proteins, glycoproteins, peptides, polysaccharides, and sometimes,lipids, pigments and metabolites. They constitute collectively the 'calcifyingmatrix', from which proteins and glycoproteins are the most studied.
In the last decade, the coupling of high-throughput screening techniques(transcriptomics + proteomics) has allowed the identification of a large numberof proteins of the "skeletal repertoires", in diverse metazoan phyla.To give an idea, in mollusks, more than 1000 proteins are now listed as putativeshell proteins, in about 30 different genera.
Proteomic data underline the diversity of these proteins, which goesalong with the diversity of functions required for calcifying a skeleton. Besideexpected members (acidic proteins, proteins with hydrophobic domains), shellproteomes (aka 'shellomes') reveal a large variety of proteins with verydifferent low complexity domains. Above all, the surprise comes from thediscovery of a huge set of proteins involved in immunity and defense mechanismsin general. This last aspect will be particularly discussed in my talk.
Dans la même collection
-
Dr Ian Dunn - The genetics and function of the cuticle, the eggs antimicrobial outer barrier.
Avian eggs have a proteinaceous cuticle covering the outside of the eggshell forming a barrier to the transmission of microorganisms. Although the cuticle is similar to the organic matrix, its
-
Dr Robbie Rae - Biological armour used to kill parasites
The co-evolution of parasites and hosts has shaped the immune system. One such ‘arms race’ is between parasitic nematodes and their molluscan hosts. One such species (Phasmarhabditis hermaphrodita)
-
Dr Claudine Blin - The innate immune function and diversity of osteoclasts
Osteoclasts are the cells responsible for bone resorption in steady state and bone destruction in chronic inflammatory diseases and osteoporosis. Up to recently, they have been considered only as
-
Dr Liliana D’Alba - Eggshell mineralization in relation to nesting ecology in reptiles
Egg morphology is essential for animal survival, mediating the interactions between embryos and their environment, and a result have evolved into an enormous diversity of forms and functions in
-
Dr Sophie Berland - Probing the mechanical properties and biochemical defence offered by shell matr…
Calcium carbonate is paired up with shell matrix proteins in the suited organo-mineral outer shell in molluscs. Advanced knowledge in the shell proteome is achieved with the development of
-
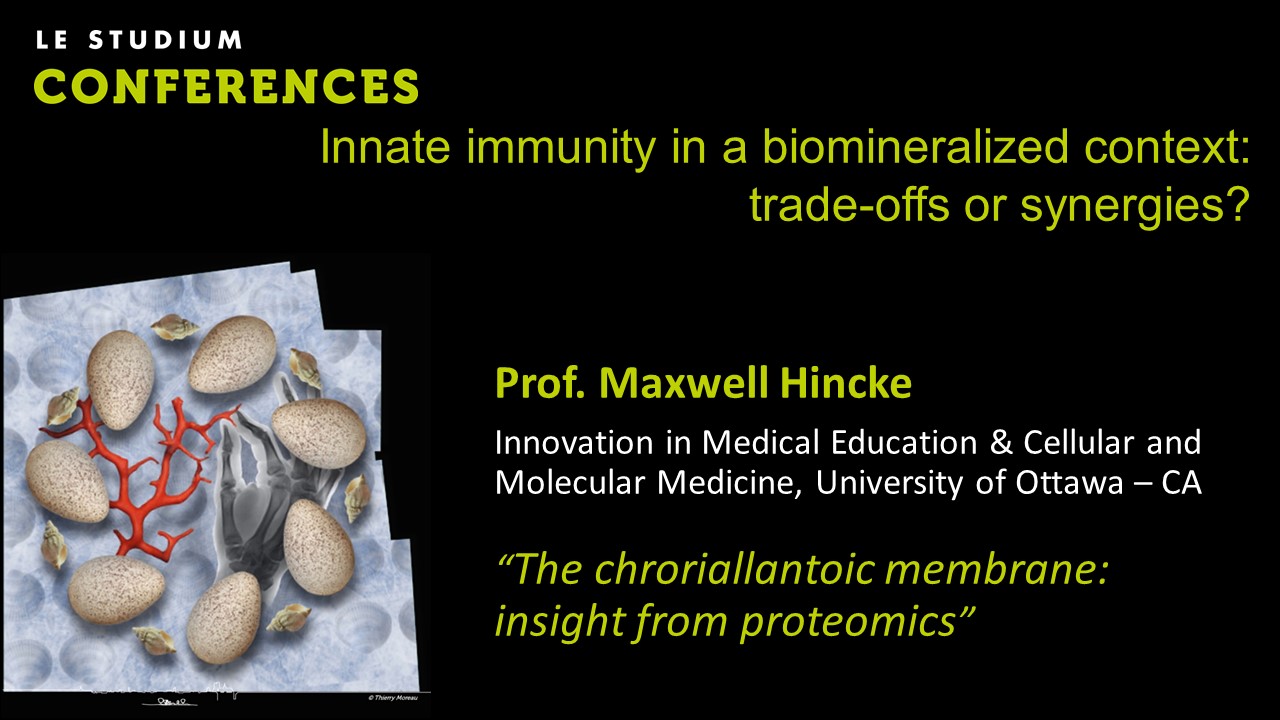
Prof. Maxwell Hincke - The chroriallantoic membrane: insight from proteomics.
In oviparous animals such as birds, embryonic development occurs in the egg; after oviposition, there is no further possibility of material exchange from the hen to fulfill the physiological needs
-
-
Prof. Marc McKee - The structure of avian (chicken) eggshell
The functional properties of biomineralized structures found in Nature result from interactions between their hybrid components – both organic (mostly proteins) and inorganic (mineral) phases – to
-
Dr Natalie Reznikov - Application of deep learning for segmentation of 3D images in biomineralizati…
Modern 3D imaging methods in biomineralization – such as X-ray tomography and dual-beam electron tomography – produce datasets that are rich in fine detail and enormous in size, often containing
-
Dr Sophie Réhault-Godbert - The eggshell microbiome
Avian eggs possess very efficient and orchestrated systems to protect the embryo during incubation, until hatch. Although the internal components of the egg are assumed to be sterile, the surface
-
Dr Christine Paillard - The Brown Ring disease in clams, a double-edged defense mechanism for shell…
Brown Ring disease is caused by Vibrio tapetis, which, by adhering to newly secreted shell matrices, interferes with the biomineralization process. Colonization and alteration of the matrices induce
-
Dr Nicolas Guyot - Phylogenetic and structural evolution of egg antimicrobial proteins and peptides
The avian egg is designed to support the autonomous development of the chick embryo in the outside environment. It initially contains all the nutrients required for embryonic growth and provides
Sur le même thème
-
Voyage au coeur des déchets électroniques
Les objets électroniques ont très largement transformé nos sociétés modernes. Ils ont permis le développement d’infrastructures toujours plus complexes et connectées, promesses d’une transition
-
Du teint de porcelaine à l'addiction solaire
par Laurence Coiffard, Laboratoire de Pharmacie industrielle et de cosmétologie, MMS EA2160, Faculté de pharmacie de Nantes.
-
21 Molecular Algorithms Using Reprogrammable DNA Self-Assembly
WoodsDamienThe history of computing tells us that computers can be made of almost anything: silicon, gears and levers, neurons, flowing water, interacting particles or even light. Although lithographically
-
-
Prof. Marc McKee - The structure of avian (chicken) eggshell
The functional properties of biomineralized structures found in Nature result from interactions between their hybrid components – both organic (mostly proteins) and inorganic (mineral) phases – to
-
Dr Natalie Reznikov - Application of deep learning for segmentation of 3D images in biomineralizati…
Modern 3D imaging methods in biomineralization – such as X-ray tomography and dual-beam electron tomography – produce datasets that are rich in fine detail and enormous in size, often containing
-
Dr Sophie Réhault-Godbert - The eggshell microbiome
Avian eggs possess very efficient and orchestrated systems to protect the embryo during incubation, until hatch. Although the internal components of the egg are assumed to be sterile, the surface
-
Dr Robbie Rae - Biological armour used to kill parasites
The co-evolution of parasites and hosts has shaped the immune system. One such ‘arms race’ is between parasitic nematodes and their molluscan hosts. One such species (Phasmarhabditis hermaphrodita)
-
Dr Nicolas Guyot - Phylogenetic and structural evolution of egg antimicrobial proteins and peptides
The avian egg is designed to support the autonomous development of the chick embryo in the outside environment. It initially contains all the nutrients required for embryonic growth and provides
-
Prof. Alejandro Rodriguez Navarro - Mechanisms of eggshell biomineralization
The avian eggshell is a thin mineral layer (350 µm thick in chicken) that protects the egg content against mechanical impacts, dehydration and microorganism contamination. Eggshell formation is a
-
Dr Sophie Berland - Probing the mechanical properties and biochemical defence offered by shell matr…
Calcium carbonate is paired up with shell matrix proteins in the suited organo-mineral outer shell in molluscs. Advanced knowledge in the shell proteome is achieved with the development of
-
Dr Ian Dunn - The genetics and function of the cuticle, the eggs antimicrobial outer barrier.
Avian eggs have a proteinaceous cuticle covering the outside of the eggshell forming a barrier to the transmission of microorganisms. Although the cuticle is similar to the organic matrix, its